What Are Mesenchymal Stem Cells?
Functions, Benefits, and Clinical Uses
Mesenchymal stem cell therapy is emerging as a powerful tool in regenerative medicine, offering new hope for treating a variety of chronic and degenerative conditions.
These multipotent stromal cells, found in tissues like bone marrow, adipose tissue, and umbilical cord, are capable of differentiating into bone, cartilage, and fat cells making them highly valuable for tissue repair and immune modulation.
Unlike embryonic stem cells, MSCs present fewer ethical concerns and carry a lower risk of immune rejection, positioning them at the forefront of modern cell-based therapy research.

What is a Mesenchymal Stem Cell?
Mesenchymal stem cells (MSCs) are a type of multipotent stromal cell capable of transforming into various tissue types, such as bone, cartilage, and fat. Originally discovered in bone marrow, these versatile cells are now sourced from other tissues including adipose tissue, umbilical cord blood, Wharton’s Jelly, and even dental pulp.
MSCs have gained significant attention in regenerative medicine for their ability to support tissue repair, reduce inflammation, and modulate immune responses without the ethical concerns associated with embryonic stem cells. Their unique biological properties make them ideal candidates for cell-based therapy, especially in orthopedic, neurological, and autoimmune disorders.
Global health organizations like the National Institutes of Health (NIH) and the International Society for Cell & Gene Therapy (ISCT) continue to support research into MSCs, driving advancements in both clinical applications and safety protocols.
Key Characteristics of Mesenchymal Stem Cells
Mesenchymal stem cells (MSCs) possess a unique combination of biological properties that make them ideal for regenerative therapies and cell-based treatments. Here’s what sets them apart:
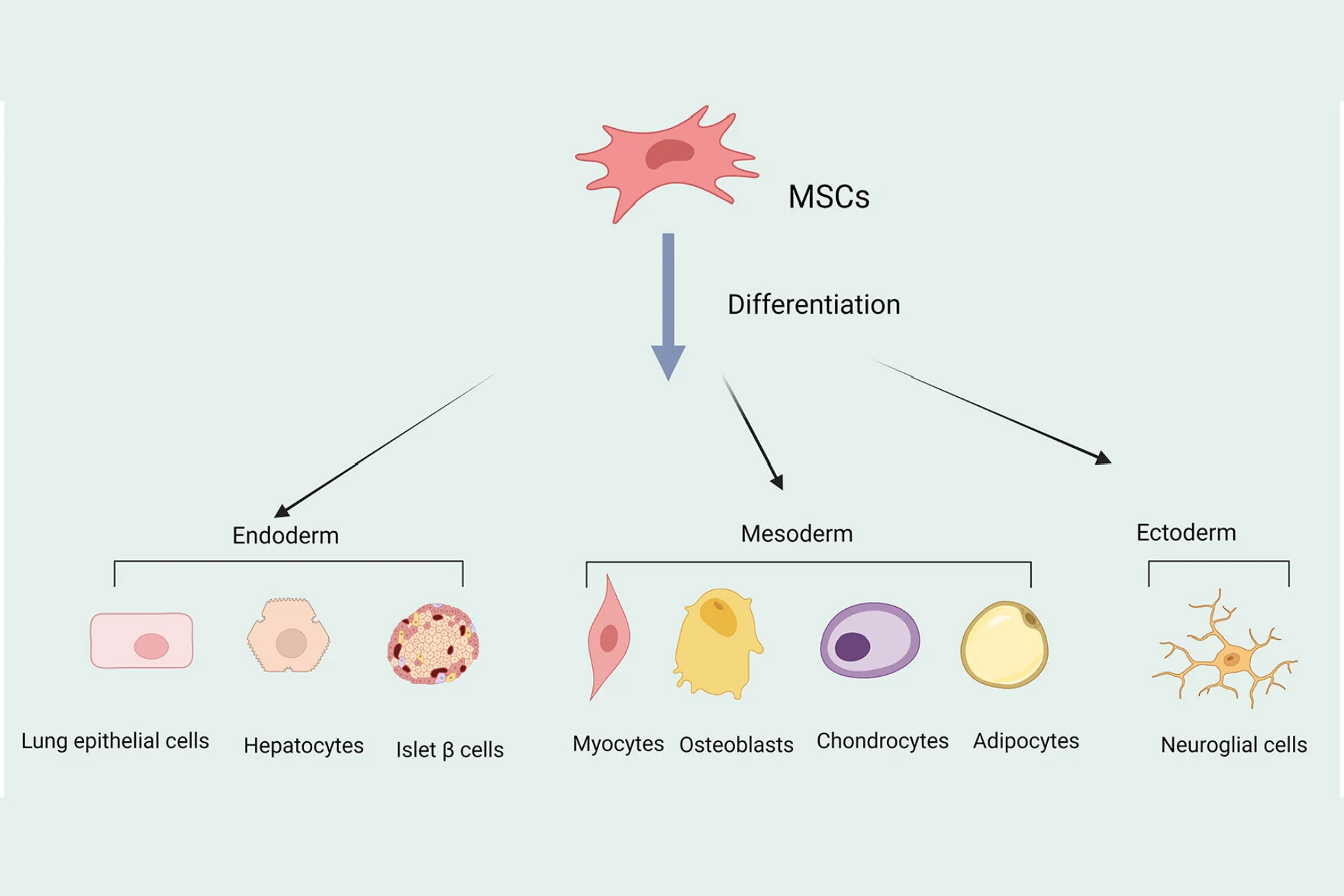
Multipotent Differentiation Ability
Mesenchymal stem cells (MSCs) are known for their ability to differentiate into multiple tissue types. They can become osteoblasts (bone cells), chondrocytes (cartilage cells), and adipocytes (fat cells), making them essential for tissue regeneration. This flexibility allows them to aid in repairing damaged or degenerated tissues, especially in orthopedic and reconstructive medicine
Self-Renewal and Plasticity
MSCs possess the capacity for self-renewal, which means they can replicate over many generations while maintaining their stem cell identity. Their plasticity and the ability to adapt to different environments supports long-term cell survival and effectiveness in therapeutic applications.
Surface Marker Expression
A key feature of MSCs is the presence of specific cell surface markers, including CD73, CD90, and CD105. These markers help researchers and clinicians identify, isolate, and verify MSC populations. Additionally, MSCs do not express hematopoietic markers like CD34 and CD45, which distinguishes them from blood-forming stem cells.
Immunomodulatory and Anti-Inflammatory Properties
One of the most important characteristics of MSCs is their role in immunomodulation. Through cell signaling, MSCs can suppress excessive immune responses and promote balance within the immune system. They secrete anti-inflammatory cytokines, such as IL-10 and TGF-β, which help reduce inflammation and support healing. These immunosuppressive properties make MSCs a promising option for treating autoimmune disorders, inflammatory diseases, and transplant rejection.
Sources of MSCs in the Human Body
Mesenchymal stem cells (MSCs) can be isolated from several tissue-specific sources throughout the body. Each source has its own advantages in terms of availability, ease of collection, and therapeutic potential. Understanding where MSCs come from helps guide their use in both autologous (patient’s own cells) and allogeneic (donor-derived) therapies.
Bone Marrow-Derived MSCs
Bone marrow was the first and most traditional source of MSCs. These cells are collected through bone marrow aspiration, often from the iliac crest. While this method is more invasive, bone marrow-derived MSCs are well-studied and often used alongside hematopoietic stem cells (HSCs) in transplantation and regenerative protocols.
Adipose Tissue-Derived MSCs
Adipose (fat) tissue offers a rich and easily accessible source of MSCs. Harvested via minimally invasive procedures like liposuction, these MSCs are abundant and highly active. Because fat is plentiful and less painful to extract, adipose-derived stem cells are frequently used in cosmetic, orthopedic, and anti-aging treatments.
Umbilical Cord
Perinatal tissues, such as the umbilical cord and Wharton’s Jelly, provide a non-invasive and ethically acceptable source of MSCs. These cells are collected at birth, making them young, highly potent, and suitable for allogeneic therapies. Their low immunogenicity makes them ideal for use across different recipients without major compatibility issues.
Placenta, Dental Pulp, and Amniotic Fluid
Additional sources include the placenta, dental pulp (from extracted teeth), and amniotic fluid. These MSCs are typically harvested during childbirth or dental procedures, offering tissue-specific MSCs for clinical or experimental use. Their applications are growing in prenatal, pediatric, and even neurological research.
Other Sources
Less commonly, MSCs can also be found in synovial fluid, peripheral blood, and menstrual blood, though these are still under early-stage investigation for mainstream use.
How Mesenchymal Stem Cells Work in the Body?
Mesenchymal stem cells (MSCs) can be isolated from several tissue-specific sources throughout the body. Each source has its own advantages in terms of availability, ease of collection, and therapeutic potential. Understanding where MSCs come from helps guide their use in both autologous (patient’s own cells) and allogeneic (donor-derived) therapies.
Paracrine Signaling and the MSC Secretome
Rather than functioning solely through differentiation, MSCs operate largely through paracrine signaling releasing a complex mix of bioactive molecules known as the secretome. This includes growth factors, anti-inflammatory agents, and anti-apoptotic proteins that help repair tissue, reduce cell death, and improve blood supply to injured areas.
Homing to Injury Sites
MSCs have the remarkable ability to home to sites of tissue damage or inflammation. Guided by chemical signals released from injured tissues, MSCs migrate through the bloodstream and localize where healing is needed. This natural targeting makes them effective in treating localized injuries without requiring direct injection into the damaged area.
Immune Response Modulation
MSCs play a critical role in regulating immune activity. They interact with immune cells such as T-cells, B-cells, and macrophages to suppress excessive immune reactions. This immunomodulatory function makes MSCs useful in treating autoimmune diseases and preventing graft-versus-host disease in transplant patients.
Cytokine Secretion and Inflammation Regulation
MSCs actively secrete anti-inflammatory cytokines such as IL-10, TGF-β, and PGE2. These molecules reduce inflammation, prevent further tissue damage, and promote a more favorable healing environment. This cytokine release also supports long-term tissue regeneration and prevents scar tissue formation in many cases.
Together, these mechanisms make MSCs powerful biological tools for managing injury, reducing inflammation, and restoring normal function. Their anti-apoptotic effects and ability to reshape the local microenvironment are what distinguish them from many other forms of therapy.
Clinical Applications of MSCs in Modern Medicine
The use of mesenchymal stem cells (MSCs) in clinical settings has grown rapidly due to their regenerative and immunomodulatory capabilities. From orthopedic injuries to autoimmune diseases, MSCs are now central to a wide range of cell-based therapies and are being actively studied in numerous trials registered on ClinicalTrials.gov. Below are some of the most promising and widely researched applications.
Orthopedic Applications
MSCs are frequently used in regenerative therapy for orthopedic conditions such as osteoarthritis, tendon injuries, and cartilage degeneration. Injected directly into joints, MSC injections promote tissue repair by reducing inflammation and stimulating cartilage regeneration. Clinical studies have shown improvements in mobility and reduced pain without the need for invasive surgery.
Neurological Conditions
In neurodegenerative diseases like Parkinson’s disease, multiple sclerosis (MS), and spinal cord injuries, MSCs are used to modulate immune responses and repair damaged neural tissue. Though still under investigation, these stem cell treatments have shown potential to improve functional recovery by reducing inflammation and encouraging nerve regeneration.
Autoimmune Diseases
MSCs are proving beneficial in treating chronic autoimmune conditions such as lupus, rheumatoid arthritis, and Crohn’s disease. Their ability to suppress immune overactivity makes them a valuable alternative to long-term immunosuppressive drugs. Trials approved under FDA regulatory oversight continue to evaluate their long-term safety and effectiveness.
Cardiovascular Diseases
For conditions like heart failure and myocardial infarction, MSCs help restore damaged heart tissue and improve blood vessel formation. Often delivered using scaffold-based therapy, MSCs promote healing of cardiac muscle and may reduce scarring and improve heart function over time.
Aesthetic and Anti-Aging Uses
In the aesthetic field, MSCs are used for skin rejuvenation, hair restoration, and scar reduction. By enhancing collagen production and tissue elasticity, MSCs offer a natural approach to anti-aging treatments. Their role in cosmetic medicine continues to expand, particularly in minimally invasive procedures.
These tissue-specific therapies powered by MSCs represent a major shift toward personalized and regenerative medicine, with clinical research accelerating globally. From the operating room to the aesthetics clinic, MSCs are reshaping how we treat damage, disease, and aging.
Benefits of Using MSCs for Therapy
Mesenchymal stem cells have several advantages that set them apart from other stem cell types, making them a preferred option in regenerative medicine and advanced cell-based therapy. Their biological properties, ease of handling, and ethical sourcing contribute to their growing clinical relevance.
Immunoprivileged with Low Immunogenicity
MSCs exhibit low immunogenicity, meaning they are less likely to trigger an immune reaction when transplanted. This immunoprivileged status reduces the risk of rejection, especially in allogeneic therapies, and allows for broader clinical use across diverse patient populations.
Simple In Vitro Expansion and GMP Compatibility
MSCs can be easily cultured and expanded in vitro while maintaining their potency. This scalability supports large-scale manufacturing under Good Manufacturing Practice (GMP) standards, enabling high-quality production for cell banking and clinical distribution.
Low Tumorigenicity and High Safety Profile
Unlike pluripotent stem cells, MSCs have a low risk of tumor formation. This safety profile makes them especially suitable for therapeutic use where long-term outcomes and patient safety are critical considerations.
Ethically Sourced and Legally Accepted
MSCs are typically derived from adult tissues or perinatal sources such as umbilical cord or placenta, making them ethical stem cells. Since their use does not involve the destruction of embryos, they are legally accepted in most countries and supported by regulatory agencies like the FDA for investigational use.
Support for Emerging Cell-Free Therapies
MSCs secrete bioactive molecules and exosomes that carry regenerative and anti-inflammatory properties. These exosomes are now being explored in exosome therapy, offering a cell-free alternative that leverages the healing power of MSCs without direct cell transplantation.
Limitations and Ethical Considerations of Mesenchymal Stem Cell Therapy
While mesenchymal stem cells (MSCs) offer tremendous promise, it’s important to recognize the limitations, risks, and ethical concerns that come with their therapeutic use. Balancing optimism with caution helps patients and practitioners make informed decisions grounded in science and regulation.
Variability Based on Age and Cell Source
The efficacy of MSC therapy can vary significantly depending on the source tissue (e.g., bone marrow vs. umbilical cord) and the age of the donor. For instance, MSCs derived from older patients often show reduced regenerative capacity and slower proliferation, which may affect clinical outcomes.
Long-Term Safety and Lack of Standardization
Despite their generally low tumorigenicity, long-term safety data for MSCs in large populations is still limited. Additionally, the lack of consistent MSC standardization such as differences in isolation methods, dosage, and cell quality poses challenges for both clinicians and researchers. These inconsistencies complicate outcome comparisons and hinder progress toward universal treatment protocols.
Regulatory and Clinical Trial Challenges
FDA regulations and other global frameworks require rigorous testing of MSC-based treatments through phased clinical trials. However, navigating this regulatory landscape is complex and costly. Many promising MSC therapies remain in investigational stages due to the time and resources needed to meet safety and efficacy standards.
Proliferation of Unregulated Clinics and Misinformation
A growing number of unregulated stem cell therapy clinics promote unproven treatments directly to patients, often bypassing clinical trial requirements. These providers pose serious therapy risks, as patients may receive unsafe or ineffective therapies without proper oversight. This also fuels public misinformation and undermines legitimate scientific progress.
Ethical Concerns and Patient Eligibility
Although MSCs avoid the ethical controversies of embryonic stem cells, questions still arise around patient eligibility, informed consent, and equitable access to treatment. Additionally, ethical debates continue about using MSCs in enhancement procedures versus life-saving interventions.
By understanding these limitations and adhering to regulated, research-backed protocols, the medical community can maximize the potential of MSCs while protecting patient safety and scientific integrity.
Current Research and Future Outlook for Mesenchymal Stem Cells
Ongoing research into mesenchymal stem cells continues to push the boundaries of what’s possible in translational medicine. With significant support from organizations like the NIH and numerous active studies on ClinicalTrials.gov, MSCs are at the center of next-generation therapies. Here’s what the future holds:
Stem Cell Banking and Global Accessibility
As MSC-based therapies become more common, stem cell banking is gaining traction. By preserving high-quality cells at an early stage often from umbilical cord or placental sources clinicians can access potent, ready-to-use MSCs when needed. Standardized, GMP-compliant cell banking is critical for ensuring long-term viability and safety.
Genetic Modification and CRISPR-Enhanced MSCs
Advanced techniques such as CRISPR gene editing are now being used to enhance the therapeutic power of MSCs. Through cellular engineering, researchers are modifying MSCs to overexpress specific anti-inflammatory proteins or to improve targeting and survival in hostile environments, offering new hope for difficult-to-treat conditions.
Scaffold and Hydrogel-Based Delivery Systems
To improve precision and effectiveness, MSCs are being embedded into scaffold-based or hydrogel delivery systems. These structures support localized cell retention and integration, particularly in orthopedic and cardiac therapies. Combining MSCs with biomaterials enhances tissue regeneration and long-term therapeutic outcomes.
Personalized and Precision Medicine Applications
The future of MSC therapy lies in its potential for personalized medicine. Using a patient’s own stem cells or customized donor MSCs allows for tailored treatments based on genetic profiles, disease stage, and tissue type. Integration with bioprinting technologies may also enable patient-specific tissue and organ regeneration in the years ahead.
As research advances and technology evolves, mesenchymal stem cells are poised to play a central role in the future of medicine bridging the gap between current clinical practice and truly personalized, regenerative therapies.
